- Title
-
Loss of popdc3 Impairs Mitochondrial Function and Causes Skeletal Muscle Atrophy and Reduced Swimming Ability in Zebrafish
- Authors
- Sun, C.C., Chen, Z.L., Yang, D., Xiao, J.L., Chen, X.T., Peng, X.Y., Wu, X.S., Tang, C.F.
- Source
- Full text @ J Cachexia Sarcopenia Muscle
|
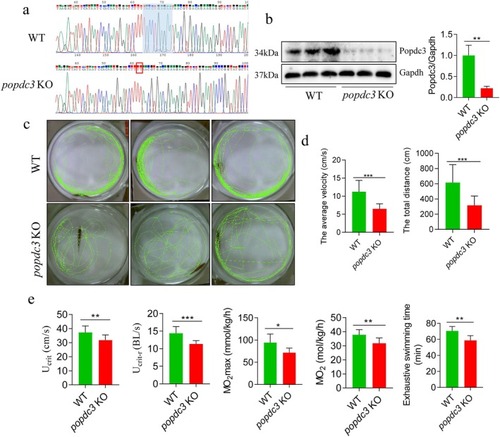
The swimming ability of |
|
Loss of |
|
|
|
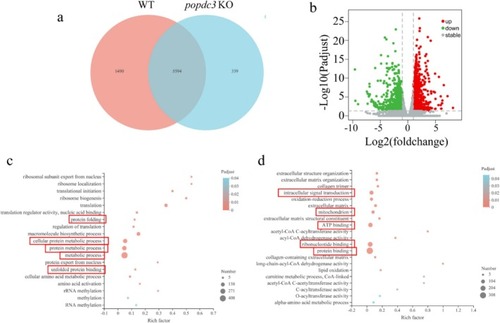
RNA‐Seq analysis of the mechanism of muscle atrophy in |
|
|
|
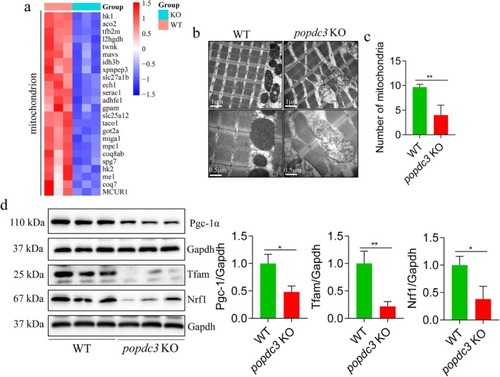
Loss of |
|
|
|
The mechanism of POPDC3 in regulating mitochondrial function and skeletal muscle mass in response to skeletal muscle atrophy. |